Solutions for the COVID-19 Fight
As the COVID-19 pandemic continues to affect our lives in many ways, there are equipment needs that have been uncovered. Here, you will find information and resources to learn about our products and services and how they can help you protect, diagnose, and treat your COVID-19 patients.
Helmer Scientific is working closely with government agencies, laboratories, pharmacies, blood banks and researchers to ensure priority access to high-quality, medical-grade equipment. Our cold storage and processing solutions are used for sample storage and processing, plasma storage and thawing, as well as COVID-19 medication storage.
Download our Vaccine Storage Readiness Guide for a complete overview of the CDC guidelines, emerging NSF standards, and how GX Solutions can help your facility rise to the challenge of safe and effective vaccine storage.
Vaccine Capacity Tool
View our interactive COVID-19 vaccine capacity tool to find the right Helmer vaccine storage equipment to meet your inventory needs.

Blood Banks and Blood Centers
COVID-19 Cold Storage and Processing Solutions for Blood Banks and Blood Centers
Laboratory Solutions
Significant steps have been taken towards efficient testing and diagnosis of COVID-19. This testing is being completed using different types of specimens, but the accuracy of all testing relies on the integrity of the samples.
Clinical specimens of suspected COVID-19 patients require cold storage to maintain their integrity. The CDC recommends storing specimens at 2-8°C for up to 72 hours after collection. If a delay in testing or shipping is expected, specimens should be stored at -70°C or below. Samples may also require long-term storage for research and development applications.
In addition, reagents used in the laboratory process require storage at refrigerated temperatures. Additional cold storage can help accommodate heightened inventory levels.
The use of GX Solutions professional, medical-grade refrigerators deliver optimized control in temperature uniformity, recovery and stability to ensure that patient samples are always stored in an optimal storage environment.
For longer term storage, Helmer Laboratory and Ultra-Low Temperature freezers, provide superior temperature uniformity and reliability along with access control options to safeguard samples.
Blood Storage and Processing Solutions
Donated plasma from patients who have recovered from COVID-19 is being used to fight the coronavirus infection. The plasma removed from recovered patients contains antibodies to the virus and is known as COVID-19 Convalescent Plasma or CCP.
Convalescent plasma (CCP) and hyperimmune globulin (hyper-IG), a blood product made from convalescent plasma are being used as therapies with the potential to lessen the severity or shorten the length of illness in patients currently fighting the infection.
These products are thawed prior to transfusion. Helmer blood bank refrigerators, plasma freezers, and plasma thawers are critical to carrying out the storage and processing of CCP.
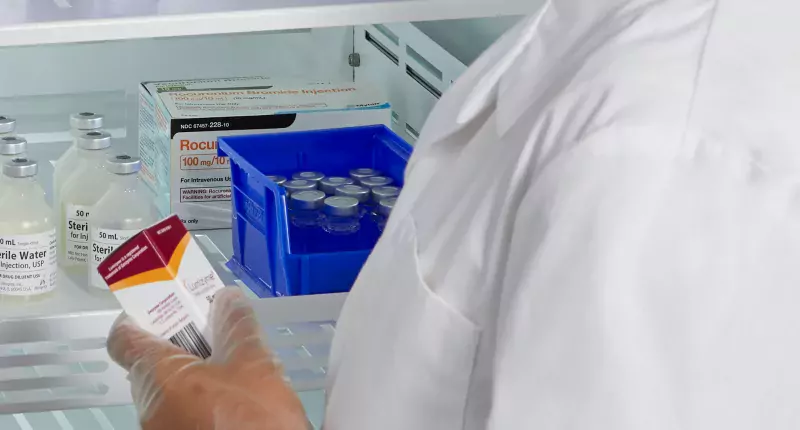
Medication and Vaccine Storage
The COVID-19 pandemic has created complex, unique challenges for refrigerated medication storage. Stress on the drug inventory is leading to conservation efforts and in some cases drug shortages. Inventory management has become crucial.
Many supportive drugs, drugs being tested for treatment and drugs undergoing clinical trials must be stored at refrigerated temperatures making storage and management of COVID-19 related therapies even more complex.
Moreover, healthcare workers need specific drugs stocked and quickly accessible in patient care areas making secure, safe, point-of-care refrigeration essential.
Helmer Scientific medical-grade refrigerators provide advanced access control and/or integration with leading ADCs via medication refrigerator locks. The BD Pyxis™ ES Refrigerator with Helmer Access Technology features precise temperature management and secure locking bins accessed only via the BD Pyxis™ MedStation™ ES.
These solutions allow drugs to be stored at refrigerated temperatures closer to the patient without compromising security or inventory control capabilities.
Medication management solutions support medication safety, clinical workflow efficiency and regulatory compliance, while helping to reduce waste and mitigate the risk of diversion.
COVID-19 Inquiries
Helmer Scientific extends a special thank you to all the healthcare providers involved in COVID-19 testing, treatment, and discovery of new therapies.
We are currently prioritizing urgent hospital needs for additional cold storage and processing equipment due to COVID-19. If you are in the process of implementing new testing sites, patient care locations, or making other preparations related to COVID-19 efforts, please let us know how we can support you.
For product availability and Immediate Ship Stock (ISS), please contact us.







